
2025年10月27日,一则震动整个医药圈的消息从美国传来:FDA正式批准了SillaJen公司基于Qureator血管化类器官模型(vTIME)疗效数据的IND申请。这是全球首个完全基于人类类器官疗效数据、无需动物药效学数据支撑的IND批准。这标志着监管大门已开,没有一只小鼠或大鼠为此“牺牲”,“人类数据优先” 的新药研发范式正式确立。
关键要素拆解:成功背后的“必要条件”此次获批并非偶然,是技术、监管与策略三者精准契合的结果。深度剖析,其成功依赖于以下几个关键要素:
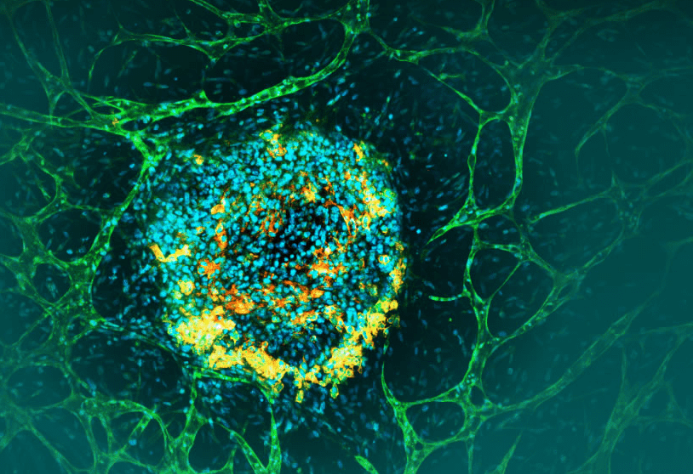
01-技术核心:超越结构的“功能性”模拟Qureator的vTIME模型提供了传统模型无法企及的数据维度:功能性血管网络:使评估药物渗透率、组织分布及靶向递送成为可能,部分替代了传统动物实验中才能获得的PK/PD数据。完整的肿瘤免疫微环境(TIME):该模型成功将人类免疫细胞纳入体系,从而能够真实模拟免疫检查点抑制剂的作用机制,并清晰展示其与靶向药BAL0891的协同效应。这正是动物模型(由于人源化免疫系统限制)的薄弱环节。AI增强的临床转化预测:集成AI引擎对多维度人类数据进行分析,旨在提升模型的临床预测价值,这无疑是增强监管机构信心的关键一环。
02-监管基础:FDA现代化法案2.0从理念到实践本次批准是《FDA现代化法案2.0》最彻底的落地。它证实了:“替代”的可行性:当人类生物学模型的数据质量足够高、维度足够全时,其证据强度可以直接等效甚至取代动物药效学实验。“人类相关性”是核心论据:申报方成功地向FDA证明了,其模型在模拟人类特异性免疫应答方面,比小鼠等动物模型具有不可替代的优越性。
03-策略选择:明智的临床与监管路径联合疗法的机制验证:选择在联合疗法(免疫+靶向)这一动物模型预测性较差的复杂领域进行验证,最大化地凸显了人类类器官模型的独特价值。与监管机构的早期和紧密沟通:从结果倒推,申报方与FDA在数据要求和标准上必然达成了前瞻性的共识。
行动指南:药企如何布局下一代临床前研究?此次获批案例,为计划采用类器官技术的药企提供了一份清晰的行动路线图。要复制这一成功路径,研发决策者应重点关注以下三个层面:
01-模型选择:从“有”到“优”,功能性与系统性是关键单纯拥有类器官已不足为奇。监管机构信服的是模型能否模拟关键生理病理过程。在选择或开发平台时,应优先考察:核心功能是否完整:例如,在代谢领域,能否模拟关键的代谢通路与毒性反应。数据维度是否丰富:平台能否提供超越细胞活性的多维数据,以构建完整的证据链。
02-监管策略:主动沟通,将NAM作为核心论证将类器官数据用于申报,需要转变与监管机构的沟通策略。早期介入:在Pre-IND会议中,就应明确提出使用NAM数据的计划,并阐述其相较于动物模型的优势和科学合理性。证据链构建:准备充分的数据,证明所选模型在模拟人类疾病生理、预测临床响应方面的可靠性与稳健性,直接回应监管机构对“人类相关性”的核心关切。
03-管线应用:精准切入,实现价值最大化在当前阶段,可优先在以下研发环节引入高仿真类器官模型,以快速验证其价值:机制验证:在靶点确认后,利用人类类器官快速验证作用机制,特别是在动物模型不敏感的领域(如免疫协同效应)。候选分子优化:在先导化合物优化阶段,用类器官数据指导分子筛选,提高候选分子的成功率。临床前到临床的转化桥梁:将类器官的药效响应数据与预期的临床生物标志物关联,为临床试验设计提供关键依据。
结语此次批准,如同第一架成功飞越大西洋的飞机。它证明了航线是通的,并详细展示了所需的飞行器规格与导航技术。这一案例清晰地指明:未来,任何旨在被监管机构采纳的类器官模型,都必须经历从适用性、局限性、相关性到可靠性、可重复性及性能标准的全面系统性验证。这一标准,正是我们始终坚守的准则。合珀生物自主研发的类器官模型,已系统完成上述维度的全面验证,其科学严谨性与数据质量完全符合FDA关于新型替代方法(NAMs)的严格要求,为将其整合进入规范化的药物研发与申报流程奠定了坚实基础。对于所有药物研发的参与者而言,一条新的航线已经开通。接下来的任务,是携手具备同样严谨技术与验证能力的伙伴,在自己的目标领域内,共同驶向更具效率与确定性的人类相关性药物研发新时代。
原文:
First FDA IND Milestone Achieved Using Human Vascularized Organoid Efficacy Data
Sponsored byQureatorOct27,20258:00am
Qureator Organ-on-Chip Breakthrough Marks Milestone Under FDA Modernization Act2.0
World’s First IND Approval in Oncology Based Solely on Human Vascularized Organoid Efficacy Data Confirms Shift Toward Human-Relevant Preclinical Testing
SAN DIEGO,Calif.–October27,2025–Qureator Inc.,a San Diego–based innovator in AI-powered,human-relevant organ-on-a-chip platforms,today announced that its proprietary vascularized tumor immune microenvironment model(vTIME)generated pivotal preclinical efficacy data that enabled SillaJen to secure U.S.Food and Drug Administration(FDA)approval of an Investigational New Drug(IND)application for a combination therapy of BAL0891with immune checkpoint inhibitors.
This landmark decision represents the world’s first FDA IND approval in which efficacy data were generated solely from human vascularized organoid–based combination studies,without relying on traditional animal efficacy(POC)testing—underscoring a fundamental shift toward human-relevant efficacy evaluation under the FDA Modernization Act2.0.
“This milestone demonstrates how close collaboration between regulators and innovators can accelerate the transition to human-relevant testing,”said Kyu Baek,Ph.D.,CEO of Qureator.“By replacing animal POC studies with more predictive,human-based efficacy models,we are reshaping how preclinical data translate into clinical outcomes.”
Transforming Preclinical Testing With vTIME
Qureator’s vTIME is a3D tumor organoid technology that accurately recreates human vascular structures and immune environments.Compared with conventional organoids,vTIME offers superior modeling of drug effects,penetration,distribution,and immune responses.Enhanced with Qureator’s Quricore AI engine,the platform integrates human data to improve clinical predictability at the preclinical stage.
In the joint study with SillaJen,a pronounced synergistic effect was observed when combining the anticancer drug BAL0891with an immune checkpoint inhibitor.The results were presented at the American Association for Cancer Research(AACR)2025Annual Meeting in April.Based on these data,SillaJen submitted an IND amendment to evaluate BAL0891with Tislelizumab(developed by BeOne Medicine,formerly BeiGene Ltd.),which the FDA approved on October6,2025.
The study was led by Dr.Sanghee Yoo,Qureator’s Head of Biology,whose team developed the vascularized tumor–immune co-culture system that generated the efficacy data supporting the IND filing.
Regulatory Momentum:Animal Efficacy Testing Phase-Out
The FDA has announced a phased reduction in animal efficacy testing and is actively promoting the use of New Approach Methodologies(NAMs).In parallel,the National Institutes of Health(NIH)has announced a new policy direction to reduce reliance on animal-only models,encouraging human-based alternatives.Together,these agencies are encouraging the industry to adopt more human-relevant approaches for preclinical decision-making.
Vascularized organoid models such as vTIME are emerging as next-generation preclinical platforms,enabling precise reconstruction of human tissue and detailed analysis of drug delivery and immune cell interactions.Industry interest has surged since the FDA’s NAM initiatives,positioning Qureator and SillaJen as early leaders in this paradigm shift.
Global Regulatory Validation
The milestone has drawn attention beyond the United States.The Ministry of Food and Drug Safety(MFDS)of Korea has also reviewed and approved Qureator’s organoid-based efficacy data,marking one of the earliest regulatory recognitions of human-relevant models in Korea.This dual validation from the FDA and MFDS reinforces Qureator’s position as a global leader in next-generation preclinical platforms.
Fruit of a Strategic Partnership
SillaJen’s proactive partnership with Qureator—initiated with a joint research agreement in June2024—was instrumental in achieving this milestone.
“Without Qureator’s collaboration,this IND approval would not have been possible,”said Seunghyun Ma,M.D.,Ph.D.,Chief Medical Officer of SillaJen.“Through their innovative vascularized organoid model,we’ve demonstrated that global regulatory agencies can approve efficacy studies without traditional animal POC testing.”
Looking Ahead:Expanding Organ-on-Chip Collaboration
Qureator plans to broaden its research collaborations to further support the FDA Modernization Act2.0.The company will showcase ongoing advancements of its vTIME platform—which utilizes human and patient-derived tissues within an organ-on-chip system—at the Society for Immunotherapy of Cancer(SITC)Annual Meeting,November5–9,2025,at the Gaylord National Resort&Convention Center in National Harbor,Md.
For more information about Qureator please visit our website at qureator.comor email us at info@qureator.com
The editorial staff had no role in this post's creation.
